GPA2 and GPB5: Dispensable or indispensable monomers or subunits of a putative heterodimer?
Main Article Content
Abstract
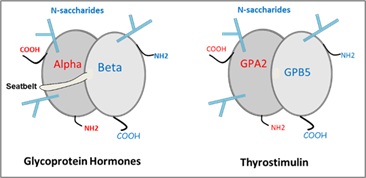
Glycoprotein hormone alpha 2 (GPA2) and glycoprotein hormone beta 5 (GPB5) have been revealed because of their homologies with glycoprotein hormone (GPH) α and β subunits respectively. Because of these homologies, GPA2 and GPB5 have been claimed to form, like GPH subunits, a non-covalent heterodimer known as Thyrostimulin, because it was found to activate the thyroid-stimulating hormone receptor (TSHR) in vitro in vertebrates. However, the real biological function(s) of GPA2 and GPB5, and whether they form functional heterodimers under physiological conditions, remain uncertain. Transcriptional and histological evidence from various species shows that GPA2 and GPB5 are most often expressed in different cell types, thus not allowing the formation of a heterodimer. When GPA2 and GPB5 are synthesized in the same cells, a GPA2/GPB5 heterodimer can form but shows very low stability. Therefore, it is more likely to act as a local paracrine or autocrine signaling rather than systemic endocrine action. In invertebrates, the gpa2 and gpb5 genes are located at the same locus without any intervening genes. Notably, the Apocrita clade appears to have lost this locus, and consequently may have lost both gpa2 and gpb5 genes. Therefore, it is of particular interest to predict the physiological consequences resulting from the absence of GPA2 and GPB5 in this group.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.